Aluminum Fluoride
Aluminum Fluoride: Anhydrous Metallurgical Grade for Electrolytic Aluminum Production
Product Name: Aluminum Fluoride
CAS No.: 7784-18-1
EINECS No.: 232-051-1
Molecular Formula: AlF₃
Molecular Weight: 83.98 g/mol
Hazardous Goods Code: 61513
UN Number: 3288
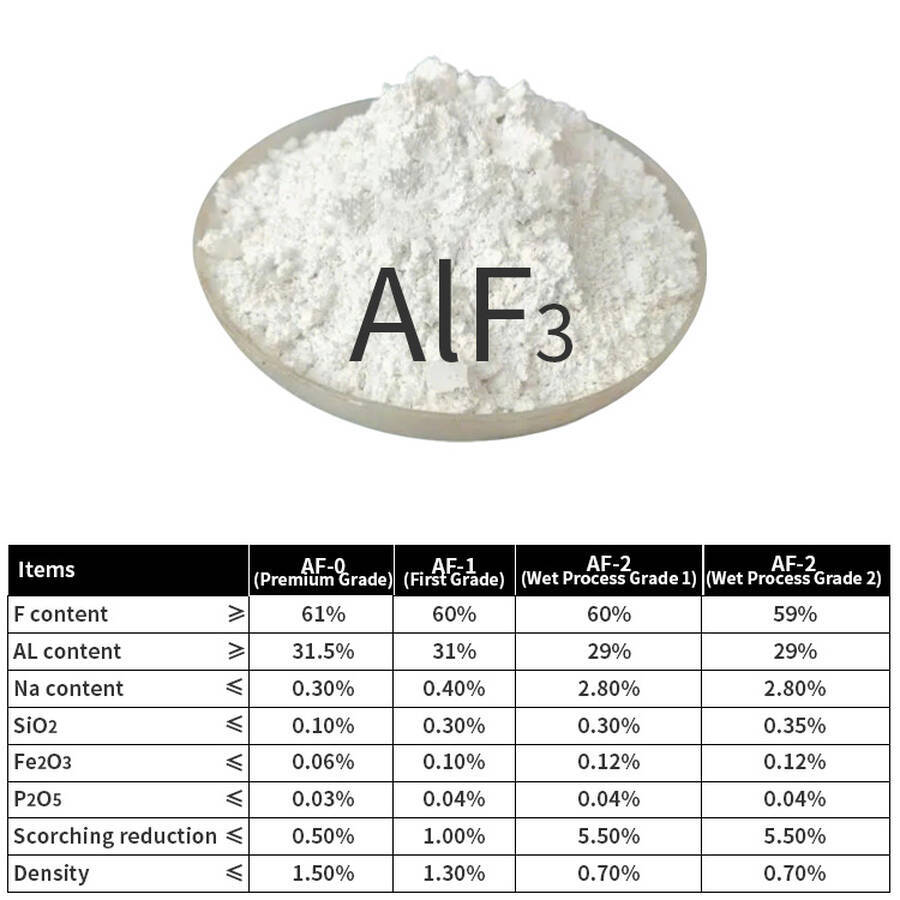
Product Classification: Anhydrous Aluminum Fluoride (AF-0, AF-1), Wet-Process Aluminum Fluoride (AF-2, AF-3), Dry-Process Aluminum Fluoride
Hazardous Chemical Classification: Class 6.1 Toxic Substance
Packaging Specification: 25 kg / 50 kg per bag
CERTIFICATES
Aluminum Fluoride: Anhydrous Metallurgical Grade for Electrolytic Aluminum Production
-
Appearance and Properties: Colorless trigonal crystals or white crystalline powder. Some products may appear transparent or translucent. No distinct odor.
-
Key Physical Parameters:
-
Relative Density (water = 1): 1.91 g/cm³
-
Melting Point: 1040°C
-
Boiling Point: 1537°C
-
Saturated Vapor Pressure: 0.13 kPa (at 1238°C)
-
Refractive Index: Approx. 1.46
-
-
Solubility: Poorly soluble in water, acids, alkalis, and most organic solvents. Can be partially hydrolyzed by water vapor at 300-400°C.
-
Chemical Stability: Stable under normal temperature and pressure. Sublimes without decomposition under strong heat. Does not react with liquid ammonia, concentrated sulfuric acid, or potassium hydroxide. Can react violently with alkali metals. Reacts chemically with strong acids.
- Core Production Processes
Process Type Raw Materials Core Process Flow Product Characteristics Anhydrous Process >99.9% Anhydrous Hydrogen Fluoride + Aluminum Hydroxide Gas-Solid Reaction Synthesis → Separation → Drying → Crushing Low hydrolysis loss, high purity, superior environmental performance Dry Process Hydrogen Fluoride Gas (from fluorite, unpurified) + Aluminum Hydroxide Gas-Solid Reaction → Crude Product Purification → Drying Moderate cost, suitable for mid-range electrolytic aluminum requirements Wet Process Aqueous Hydrofluoric Acid (from fluorite) + Aluminum Hydroxide Liquid-Solid Reaction → Precipitation → Centrifugal Separation → Drying Mature technology, suitable for applications with lower purity requirements - Main Application Areas
6.1. Electrolytic Aluminum Industry (Core Application): Serves as a key additive in the cryolite-alumina electrolyte. Lowers the liquidus temperature of the electrolyte, adjusts the bath ratio, improves electrical conductivity, optimizes the thermal balance of the electrolysis process, and ultimately enhances aluminum product quality and production efficiency.
6.2. Ceramic and Enamel Industry: Acts as a flux and component in glazes, improving product gloss, strength, melting uniformity, and glaze adhesion.
6.3. Chemical and Materials Sector: Catalyst in organic synthesis (e.g., hydrocarbon cracking, esterification). Substrate for phosphors (used with rare earths in LEDs, fluorescent lamps). Component in metal welding fluxes.
6.4. Other Uses: Flux in non-ferrous metal smelting. Secondary fermentation inhibitor in alcohol production. Refractive index modifier for optical lenses and prisms. Corrosion protection agent for aluminum alloy surfaces.
7. Safety and Protection Guidelines
7.1.Hazard Overview
-
Health Hazards: Acute Toxicity (Oral), Category 3 (Mouse LD₅₀=103 mg/kg, Rat LD₅₀=1800 mg/kg). Decomposition products (hydrogen fluoride) can irritate eyes and the respiratory tract, causing bronchitis and pneumonia. Long-term exposure may lead to skeletal fluorosis and is suspected of damaging fertility.
-
Environmental Hazards: Poses potential risks of water and soil pollution; prevent leakage and spread.
-
Reaction Risks: Non-combustible. Reacts violently with alkali metals. In a fire, can release toxic gases such as hydrogen fluoride and aluminum oxide.
7.2. Handling and Storage Requirements
-
Handling Instructions: Use enclosed operations with local exhaust ventilation. Operators must wear self-inhalation filtering dust masks, chemical safety goggles, gas-permeable protective clothing, and latex gloves. Avoid generating dust. Strictly prohibit storage or handling with acids. Handle with care during transport.
-
Storage Conditions: Store in a cool, ventilated warehouse. Temperature should not exceed 30°C, relative humidity ≤80%. Keep packaging sealed. Store separately from acids, edible chemicals, and alkali metals. The storage area should be equipped with leak containment tools and dust collection equipment.
7.3. Emergency Response Measures
-
Spill Response: Isolate the contaminated area and restrict access. Emergency personnel should wear full-face dust masks and protective clothing. Avoid raising dust. For small spills, collect carefully with a clean shovel into a sealed container. For large spills, cover with plastic sheeting for later recovery and disposal.
-
First Aid Measures:
-
Skin Contact: Remove contaminated clothing immediately. Wash skin thoroughly with soap and water and flowing water for 15 minutes. Seek medical attention.
-
Eye Contact: Lift eyelids and rinse continuously with physiological saline or flowing water for 10-15 minutes. Seek immediate medical attention.
-
Inhalation: Move person to fresh air. Administer oxygen or artificial respiration if necessary. Seek medical attention.
-
Ingestion: Rinse mouth and drink warm water. Do NOT induce vomiting. Immediately call a poison control center or doctor.
-
-
Fire Fighting: Firefighters must wear full-body protective clothing and fight fire from upwind. Use large amounts of water fog to extinguish fire and cool containers, while dispersing toxic gases.
7.4. Transportation Requirements
-
Packaged in woven bags with plastic liner. Net weight: 25kg/50kg per bag.
-
Strictly prohibit mixed transport with acids, oxidizing agents, food, and food additives.
-
Protect from direct sunlight, rain, and high temperatures during transport. Ensure cargo hold is ventilated. Equip vehicles with leak emergency response equipment.