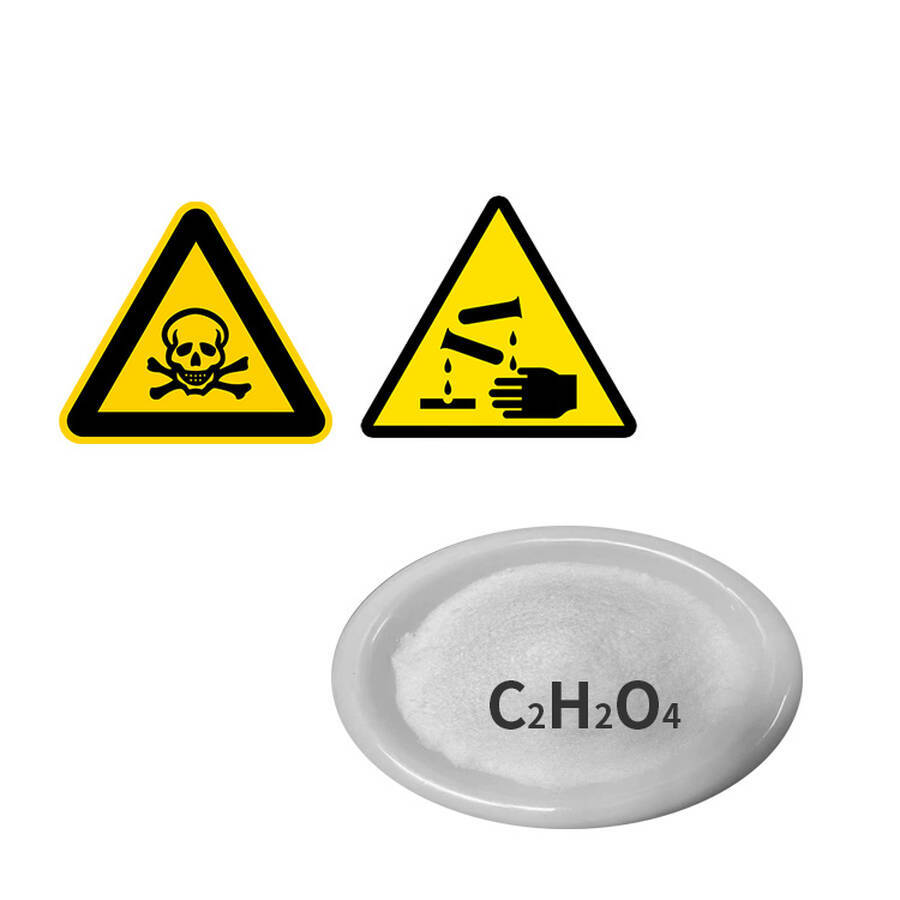
C₂H₂O₄ Oxalic Acid
Wholesale Buy Oxalic Acid Anhydrous Ethanedioic Acid Price for Sale
Chinese Name: Oxalic Acid (Ethanedioic Acid)
Assay (Purity): 99.99%
CAS No.: 6153-56-6 (Note: Common anhydrous form is 144-62-7)
EINECS No.: 205-634-3
Molecular Formula: C₂H₂O₄
Molecular Weight: 90.04 (Anhydrous)
Hazard Identification Number: 81614
UN Number: 3261
Hazard Classification: Class 8 Corrosive Substance (Acidic)
CERTIFICATES
C₂H₂O₄ Oxalic Acid
I. Physicochemical Properties
-
Appearance & Characteristics
-
Anhydrous: White crystalline powder or columnar crystals, hygroscopic, absorbs moisture from air.
-
Industrial grade may appear light gray or pale yellow due to trace impurities, without affecting performance.
-
-
Key Physical Parameters
-
Relative Density (water = 1, 20°C): 1.90 g/cm³
-
Melting Point: 189.5°C (decomposes to CO, CO₂ and H₂O)
-
Solubility: Highly soluble in water (14.3g/100mL at 20°C); soluble in ethanol and ether; slightly soluble in benzene and chloroform.
-
Aqueous Solution: Strongly acidic (pH ≈1.3 for 0.1 mol/L solution).
-
-
Chemical Stability
-
Stable at room temperature; exhibits reducibility and corrosiveness.
-
Reacts with strong oxidizers (e.g., potassium permanganate), metals (e.g., iron, zinc), and bases.
-
Decomposes at high temperatures, releasing toxic carbon monoxide.
-
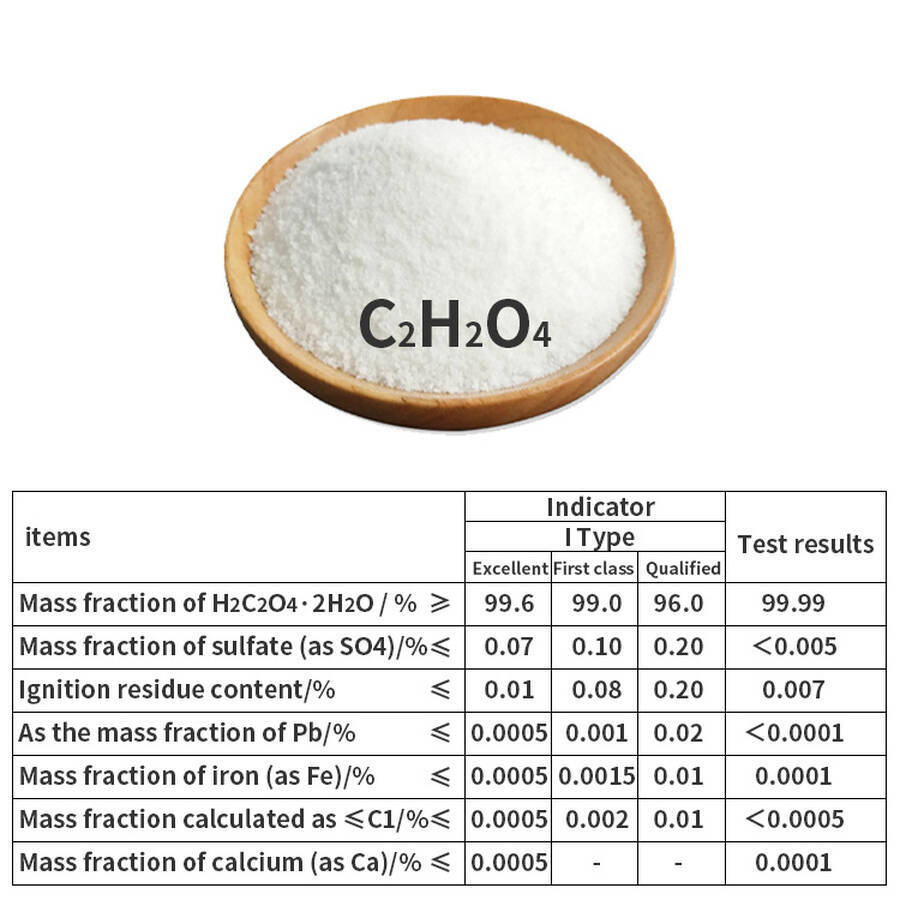
II. Quality Standards (Industrial Grade, GB/T 1626-2008)
| Parameter | Premium Grade | First Grade | Qualified Grade | Test Result |
|---|---|---|---|---|
| Oxalic Acid Content (%) | ≥99.6 | ≥99.0 | ≥96.0 | 99.99 |
| Sulfate (as SO₄) (%) | ≤0.07 | ≤0.10 | ≤0.20 | <0.005 |
| Residue on Ignition (%) | ≤0.01 | ≤0.08 | ≤0.20 | 0.007 |
| Heavy Metals (as Pb) (%) | ≤0.0005 | ≤0.001 | ≤0.02 | <0.0001 |
| Iron (as Fe) (%) | ≤0.0005 | ≤0.0015 | ≤0.01 | 0.0001 |
| Chloride (as Cl) (%) | ≤0.0005 | ≤0.002 | ≤0.01 | <0.0005 |
| Calcium (as Ca) (%) | ≤0.0005 | — | — | 0.0001 |
III. Main Application Fields
-
Industrial Applications (>80% usage):
-
Mining: Extraction and separation of rare earth elements.
-
Metal Processing: Rust and oxide removal; metal polishing.
-
Textile Industry: Bleaching agent and mordant for cotton/linen.
-
Chemical Synthesis: Production of oxalates, pharmaceuticals, and pesticides.
-
Leather Industry: Tanning agent for improved softness and durability.
-
-
Other Fields:
-
Environmental: Wastewater treatment for metal ion removal and chromium reduction.
-
Laboratory: Analytical reagent and culture medium preparation.
-
Household: Cleaning agent for descaling and stain removal.
-
IV. Safety & Protection Guidelines
(I) Hazard Overview
-
Health Hazards: Toxic and corrosive. Oral lethal dose ~15–30g for adults. Causes skin/eye damage, respiratory irritation, and organ failure if ingested.
-
Environmental Risks: Acidifies water; forms stable complexes with metal ions.
-
Fire/Explosion: Non-flammable but reacts vigorously with oxidizers; decomposes to CO at high temperatures.
(II) Handling & Storage
-
Operations:
-
Use acid-resistant gloves (nitrile), goggles, chemical suits, and dust masks.
-
Always add acid to water slowly with stirring. Ensure ventilation.
-
Separate from oxidizers, alkalis, and metal powders.
-
-
Storage:
-
Cool (≤30°C), dry, ventilated area; sealed PE plastic or lined steel drums.
-
Separate from foodstuffs, oxidizers, and alkalis; provide spill containment.
-
(III) Emergency Measures
-
Spills: Isolate area. Neutralize with sand/lime; collect in corrosion-resistant containers.
-
First Aid:
-
Skin/Eyes: Flush with water for 15 min; seek medical care.
-
Inhalation: Move to fresh air; administer oxygen if needed.
-
Ingestion: Drink milk/egg white; DO NOT induce vomiting.
-
-
Firefighting: Use water spray; wear full protective gear. Employ dry powder/CO₂ for oxidizer-related fires.
(IV) Transportation
-
Packed in woven bags with inner liner.
-
Use certified corrosive-resistant vehicles; never mix with food/oxidizers/alkalis.
-
Avoid sunlight, rain, and high temperatures; carry neutralizers and emergency tools.
Feel free to inquire anytime and lock in your favorite style!