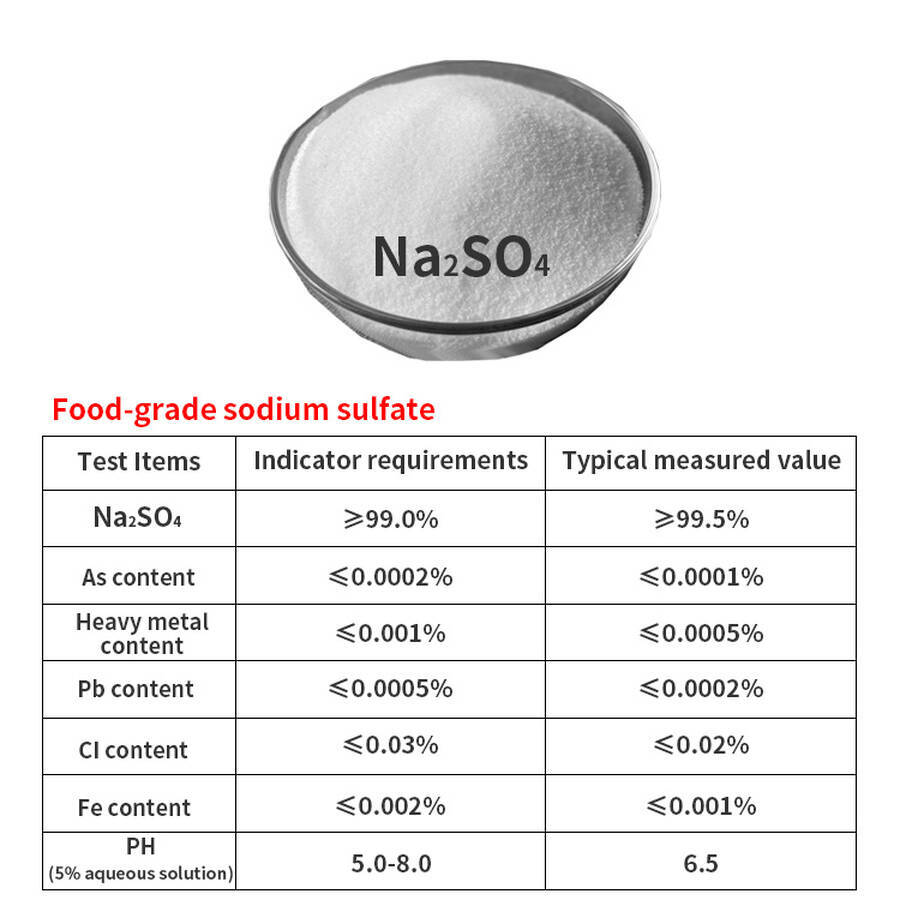
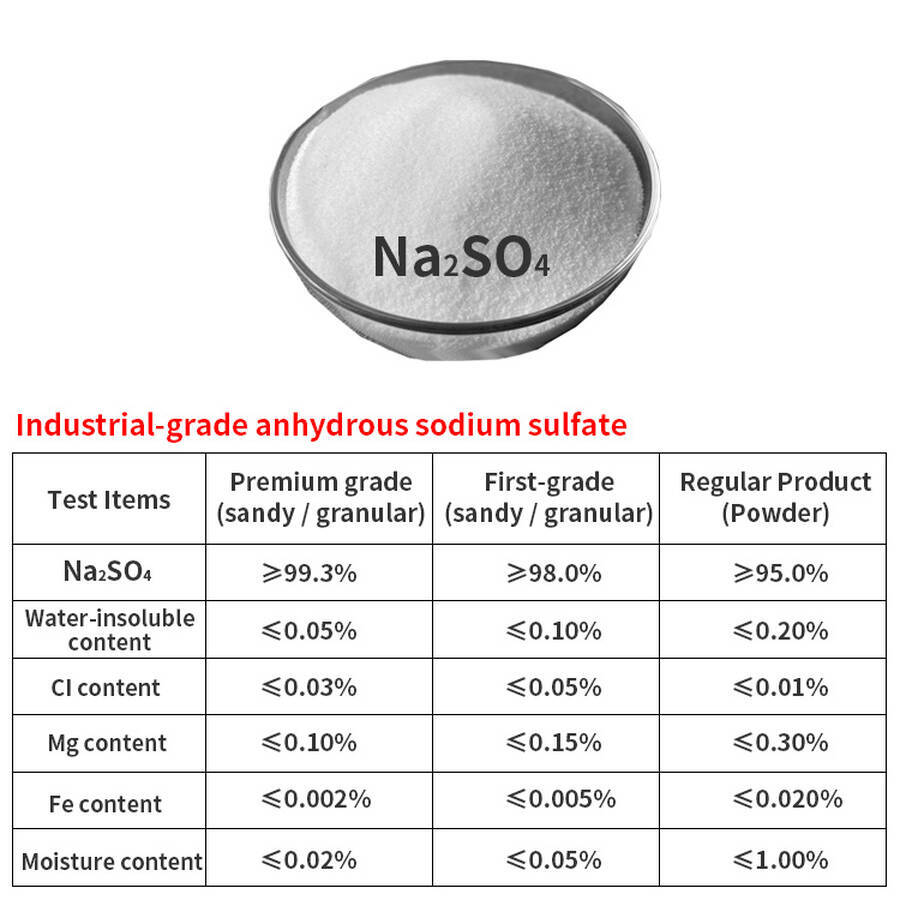
Sodium Sulphate Na₂SO₄
High-Purity Anhydrous & Decahydrate Crystals for Glass, Detergent & Chemical Industries
Product Name: Sodium Sulphate
Molecular Formula: Na₂SO₄
CAS No.: 7757-82-6
EINECS No.: 231-820-9
Molecular Weight: 142.04 g/mol
Hazardous Goods Code: None (Non-hazardous chemical under normal conditions. Certain high-purity industrial grades are regulated as general cargo.)
UN Number: None (Non-hazardous goods)
Product Classification: Anhydrous Sodium Sulfate By Application: Industrial Grade Sodium Sulfate, Food Grade Sodium Sulfate, Pharmaceutical Grade Sodium Sulfate By Purity: Premium Grade, First Grade, Qualified Grade By Production Process: Mineral, By-product
Packaging Specification: Ton
CERTIFICATES
High-Purity Anhydrous & Decahydrate Crystals for Glass, Detergent & Chemical Industries
1. Appearance and Properties:
-
Anhydrous Sodium Sulfate: White crystalline powder or granules. Odorless and tasteless. Hygroscopic; readily absorbs moisture from air leading to caking.
2. Key Physical Parameters:
-
Anhydrous Sodium Sulfate:
-
Relative Density (water = 1, 20°C): 2.68 g/cm³
-
Melting Point: 884°C
-
Boiling Point: 1430°C (with decomposition)
-
-
Solubility: Both anhydrous and crystalline forms are readily soluble in water. Solubility in water is approximately 19.5 g/100 mL at 20°C. The aqueous solution is neutral (pH ≈ 7). Slightly soluble in ethanol; insoluble in acetone, diethyl ether, and other organic solvents.
3. Chemical Stability:
Stable under normal temperature and pressure conditions. Exhibits no oxidizing or explosive properties. Does not react violently with strong acids or strong bases. At high temperatures, it can react with carbon to form sodium sulfide and sodium carbonate. Reacts with barium salts and lead salts to form insoluble sulfate precipitates. Can form double salts with metal ions (e.g., Ca²⁺, Mg²⁺).
4. Sodium Sulfate Production Processes
|
Raw Materials | Core Process Flow | Product Characteristics | |
| Natural Mineral Extraction | Mirabilite ore, Thenardite ore | Ore Mining → Crushing → Dissolution (or Direct Crystallization) → Filtration & Purification → Evaporation & Concentration → Cooling Crystallization (or Drying & Dehydration) → Screening → Finished Product | Low production cost; Product purity is influenced by ore quality; Suitable for large-scale production of industrial-grade sodium sulfate. | |
| Chemical By-product Recovery | By-products from sulfate process TiO₂ production, Waste liquors from chlor-alkali industry, etc. | By-product Collection → pH Adjustment via Neutralization → Filtration & Purification (removal of impurities e.g., titanium, iron) → Evaporation & Concentration → Crystallization → Drying → Refining → Finished Product | Enables resource recycling; Relatively high purity; Can be further purified to food or pharmaceutical grade based on requirements. | |
| Chemical Synthesis | Sodium Hydroxide, Sulfuric Acid (or Ammonium Sulfate) | Controlled Reaction of sodium hydroxide solution with sulfuric acid (or ammonium sulfate) → Neutralization to pH 6-7 → Filtration & Purification → Evaporation & Concentration → Crystallization → Drying → Finished Product | High product purity (can reach ≥99.5%); Controllable process; Suitable for high-purity applications like food and pharmaceutical grades. |
5. Main Application Areas
5.1. Industrial Sector (Core Applications):
-
Detergent Industry: Used as a filler and detergent builder in laundry powder and liquid detergents. Enhances cleaning power and reduces surfactant dosage.
-
Textile Industry: Serves as a levelling agent in cotton dyeing to improve dye uptake. Also used as a scouring agent for silk and wool fabrics to remove impurities.
-
Paper Industry: Acts as a cooking agent in pulping to improve pulp quality. Used to regulate pH during the paper sizing process, enhancing paper strength.
-
Glass Industry: Functions as a fluxing agent, lowering the melting temperature of glass and reducing energy consumption. Also helps suppress bubble formation in the glass melt, improving transparency.
5.2. Food and Pharmaceutical Sector:
-
Food Additive: Used as a processing aid in baked goods (to adjust dough viscosity), beverages (as a stabilizer), and canned products (as a quality improver), within the limits specified by GB 2760.
-
Pharmaceutical Industry: Pharmaceutical-grade sodium sulfate (Glauber's Salt) can be used as a laxative to relieve constipation. Also used in preparing medical formulations like saline solutions and disinfectants, or as an excipient to adjust osmotic pressure.
5.3. Other Sectors:
-
Agriculture: Used as a soil amendment for saline-alkali land to reduce soil salt concentration and improve soil structure. Applied in small quantities as a foliar fertilizer to supplement sulfur and sodium.
-
Chemical Synthesis: Used in the production of various chemicals, including sodium silicofluoride, sodium sulfide, glass refining agents, sodium silicate, and detergent intermediates.
-
Environmental Protection: Employed in wastewater treatment to remove calcium and magnesium ions, reducing water hardness. Also used as a coagulant aid in treating wastewater containing heavy metals.
6. Safety and Protection Guidelines
6.1. Hazard Overview
-
Health Hazards: Generally exhibits low toxicity upon normal contact. Inhalation of dust may irritate the respiratory tract, causing coughing and sneezing. Prolonged skin contact with dry powder may cause mild dryness and flaking. Ingestion of large amounts (non-food-grade) may cause gastrointestinal discomfort, such as nausea and diarrhea (food-grade poses no risk when used according to standards).
-
Environmental Hazards: Excessive release into water bodies may cause eutrophication, affecting aquatic life. However, it is biodegradable and can be gradually diluted and degraded in the environment, exhibiting no long-term cumulative toxicity.
-
Fire & Explosion Risk: Non-flammable, non-explosive. No hazardous reaction upon exposure to open flame or high temperature. No fire or explosion risk when mixed with strong oxidizing agents.
6.2. Handling and Storage Requirements
-
Handling Instructions:
-
When handling powdered products, wearing a dust mask (e.g., N95) is recommended to avoid dust inhalation. Wash hands after contact. Maintain ventilation in the operating area.
-
For dissolution, slowly add sodium sulfate to water with stirring until dissolved; no special temperature control is required (the dissolution of decahydrate may involve slight heat absorption, which is normal).
-
Avoid prolonged heating with strong acids (e.g., sulfuric acid) to prevent the generation of irritating gases. Separate operations from barium and lead salts to avoid the formation of toxic precipitates.
-
-
Storage Conditions:
-
Store in a cool, dry, well-ventilated warehouse, avoiding direct sunlight. Anhydrous sodium sulfate must be protected from moisture to prevent caking. Sodium sulfate decahydrate must be protected from high temperatures (above 32°C) to prevent efflorescence (loss of water of crystallization).
-
Packaged in polyethylene woven bags (with plastic liner) or paper bags. Net weights: 25kg/50kg/1000kg. Separate storage is required for food-grade, industrial-grade, and pharmaceutical-grade products; mixing is strictly prohibited.
-
Keep away from food raw materials, pharmaceuticals, and strongly corrosive substances. Regularly inspect packaging integrity in the storage area; replace damaged packages promptly.
-
6.3. Emergency Response Measures
-
Spill Response:
-
Small Spills: Collect powder using a clean broom or shovel and place in sealed bags for industrial recycling or compliant disposal.
-
Large Spills: Cover with plastic sheeting to prevent dust dispersion. Collect gradually into corrosion-resistant containers. Dispose of via professional organizations. Prevent entry into sewers or rivers. If entry into a water body occurs, notify environmental authorities promptly.
-
-
First Aid Measures:
-
Skin Contact: Remove contaminated clothing. Rinse skin with flowing water for 1-2 minutes. Apply moisturizer; no specific treatment is usually required.
-
Eye Contact: Immediately open eyelids and rinse thoroughly with physiological saline or flowing water for 5-10 minutes. If discomfort persists, seek medical attention.
-
Inhalation: Move to fresh air. Keep the respiratory tract clear. If coughing or chest tightness occurs, rest usually provides relief. Seek medical attention for severe symptoms.
-
Ingestion (Non-Food-Grade): Immediately drink plenty of warm water for dilution and expulsion. If vomiting or diarrhea occurs, seek medical attention (no treatment needed for food-grade if ingested according to standards).
-
-
Fire Fighting Methods: The substance itself is non-combustible. No special firefighting measures are required. Focus on cooling surrounding containers to prevent packaging damage and product release due to heat. If a fire involves other combustible materials, use water, dry powder, or carbon dioxide extinguishers.
6.4. Transportation Requirements
-
Packaging must use sturdy plastic woven bags with tight seals to prevent leakage and moisture absorption during transport. Food-grade product packaging must be marked with "Food Additive" and "Keep Dry" labels.
-
Vehicles do not require hazardous goods transport qualifications. Compartments must be kept dry and clean. Avoid mixed transport with damp goods or corrosive substances.
-
Protect from direct sunlight, rain, and high temperatures during transport. Handle with care during loading/unloading to prevent packaging damage. For long-distance transport of anhydrous sodium sulfate, desiccants can be placed in the compartment to prevent caking.