Na2SiF6 Sodium Fluosilicate
Common All Inorganic Compounds Na2SiF6 Sodium Fluosilicate
Chinese Name: Sodium Hexafluorosilicate
Common Aliases: Sodium fluorosilicate, Sodium hexafluorosilicate
CAS No.: 16893-85-9
Molecular Formula: Na₂SiF₆
Molecular Weight: 188.06
Hazard Identification Number: 61514
UN Number: 2674
Hazard Classification: Class 6.1 Toxic Substances
Place of Origin: Yunnan, China
CERTIFICATES
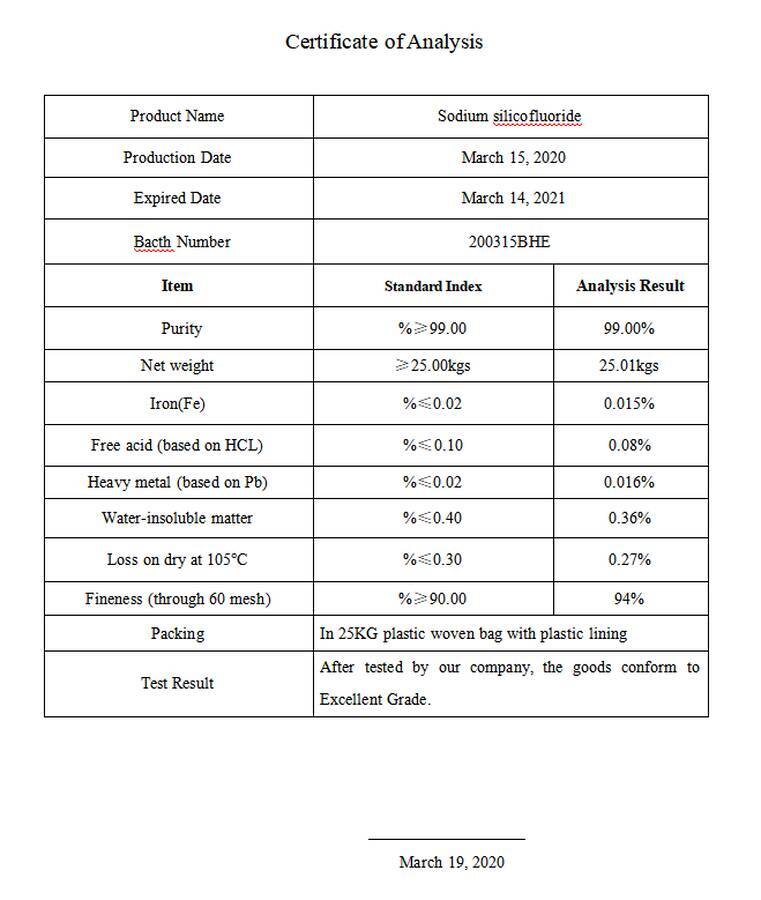
$200315BHE
Certificate of Analysis
Na2SiF6 Sodium Fluosilicate
I. Physicochemical Properties
-
Appearance: White crystalline powder, odorless, tasteless, hygroscopic.
-
Density: Relative density (water = 1) approx. 2.68–2.75 g/cm³.
-
Melting & Decomposition: Melting point 847°C; decomposes above 300°C into sodium fluoride (NaF) and silicon tetrafluoride (SiF₄) gas.
-
Solubility: Slightly soluble in water (1 part in 150 parts cold water, 1 part in 40 parts boiling water); insoluble in ethanol; soluble in ether. Aqueous solution is neutral in cold water and turns acidic upon decomposition in hot water.
-
Stability: Stable under normal temperature and pressure. Reacts with acids to release corrosive and irritating hydrogen fluoride and silicon tetrafluoride gases. Non-combustible, non-explosive.
II. Main Applications
-
Glass & Enamel Industry: Opacifier in glass production; fluxing agent in enamel manufacturing.
-
Water Treatment: Fluoridation of drinking water to enhance safety.
-
Mineral Processing: Used primarily as a depressant to inhibit the floatability of specific minerals (especially silicates), enabling separation of target minerals from gangue.
-
Construction Industry: Setting agent in acid-resistant mortar and concrete; hygroscopic agent in acid-resistant cement.
-
Preservation & Protection: Wood and leather preservative; coagulant for natural latex products.
-
Agriculture & Chemical Industry: Raw material for agricultural insecticides; intermediate in the production of other fluorides (e.g., sodium fluoride, cryolite); additive in ore refining.
-
Other Fields: Pharmaceutical excipient; rubber industry additive; silver smelting and processing, etc.
III. Quality Standards(Complies with National Standard GB/T 23936-2018)
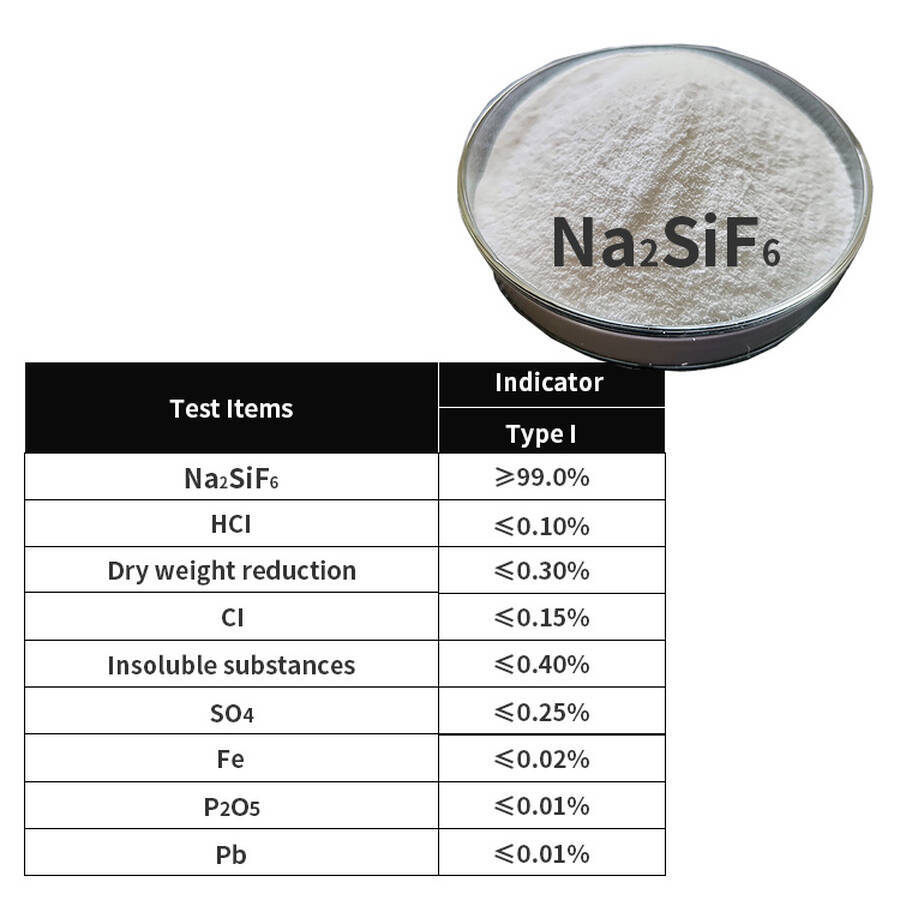
| Project | Type I Premium Grade |
| Sodium Fluorosilicate (Na₂SiF₆) ω/% | 99.0 |
|
Free acid (as HCl)/% |
0.10 |
|
Loss on drying ω/% |
0.30 |
|
Chlorides (as Cl)/% |
0.15 |
|
Water-insoluble matter ω/% |
0.40 |
|
Sulfates (as SO₄)/% |
0.25 |
|
Iron (Fe)/% |
0.02 |
|
Phosphorus pentoxide (P₂O₅)/% |
0.01 |
|
Heavy metals (as Pb)/% |
0.01 |
IV. Safety & Protection Guidelines
(I) Hazard Overview
-
Health Hazards: Moderately toxic. Oral LD₅₀ in rats: 125 mg/kg. Ingestion may cause nausea, vomiting, bloody vomit/diarrhea; severe cases may lead to convulsions, shock, or death. Skin contact may cause dermatitis or cracking.
-
Environmental Hazards: Risk of water pollution; avoid leakage into water bodies.
-
Fire & Explosion Hazard: Non-flammable. Reacts with acids to release toxic gases.
(II) Handling & Storage Requirements
- Handling: Enclosed operation with local exhaust ventilation. Operators must wear self-priming filter dust masks, chemical safety goggles, anti-permeation workwear, and latex gloves. Avoid dust generation; keep away from oxidizers; handle with care.
- Storage: Store in a cool, well-ventilated area away from fire and heat sources. Separate from oxidizers and food chemicals. Provide leak containment materials in storage area.
(III) Emergency Measures
-
Leak Handling: Isolate contaminated area. Responders must wear full-face dust masks and protective clothing. For minor leaks, sweep up carefully and transfer to bags. For major leaks, cover with plastic sheeting for recovery.
-
First Aid:
-
Skin contact: Remove contaminated clothing, rinse with flowing water.
-
Eye contact: Lift eyelids, rinse with water or saline; seek medical attention.
-
Inhalation: Move to fresh air, administer oxygen, seek medical care.
-
Ingestion: Drink plenty of warm water to induce vomiting; seek immediate medical help.
-
-
Firefighting: Firefighters should wear full-body protective suits. Fight fire from upwind. Move containers to open area if safe. No special extinguishing agents required.
(IV) Transportation Requirements
-
Packed in woven bags with inner plastic liners.
-
Do not transport with acids, oxidizers, food, or food additives.
-
Protect from direct sun, rain, and high temperatures during transit. Equip with leak emergency response equipment.
Feel free to inquire anytime and lock in your favorite style!